Research Article - (2021) Volume 6, Issue 3
Assessment the Average Age of Menarche and its Relationship to Weight, Height and Body Mass Index of Adolescent Girls in North-East of Iran
Hossain Nassirian1, Saeedeh TarvijEslami1*, Hamed Shafagh1, Maryam Gholami2 and Kimia Vatani1Abstract
Purpose: Anthropometric characteristics are partly related to menarche age. We aimed to survey the age of menarche, and its relationship to weight, height and Body Mass Index in adolescent girls.
Methods: In this cross sectional study, 460 students aged 18-21 years old at Mashhad University of Medical Sciences included. Data was obtained through questiones. BMI>30 kg/m2 was considered as obesity and a menarche age of less than 11 years was considered as early menarche.
Results: The mean age of menarche was 13.32 years, in students with BMI less than 30 kg/ m2. 13.8% of the girls had menarche age of lower than 11 years. There was a significant relationship between history of age at menarche and current height, p<0.05 with no significant association between age of menarche and the current weight and BMI, (p>0.05). Age at menarche was slightly earlier in obese girls but not significantly.
Conclusions: The earlier the menarche, the shorter the final height, P<0.05. There was no significant association between age at menarche and obesity. More studies should be performed to survey in this field to enhance families care for promotion of height and health of girls.
References
Stone Market in Florida Stone Market in Newyork Stone Market in Alabama Stone Market in Miami Stone Market in Los AngelesKeywords
Height, Weight, Menarche, Body mass index, Medical students
Introduction
Between early childhood and approximately 8-9 yrs of age (prepubertal stage), the hypothalamic pituitary gonadal axis is dormant, as reflected by undetectable serum concentrations of LH and sex hormones. In females, the breast bud (thelarche) is usually the first sign of puberty (10-11 yrs of age and some showing as early as age seven years), followed by the appearance of pubic hair (pubarche), 6-12 months later. The interval to the onset of menstrual activity (menarche) is usually 2-2.5 yrs, but may be as long as 6 yrs. Peak height velocity occurs early between 11 and 12 yrs of age in girls and always precedes the menarche. The mean age of menarche is approximately 12.75 yrs [1]. During the past decades, the age of onset of puberty in different populations has fallen. The age of breast development has been dropping much faster than the age of menarche [2]. In Western Europe, between 1850 and 1960, age of menarche per decade, was reduced to 4 months per decade [3-5]. Children with low age of menarche are at risk of accelerated skeletal maturation and short stature in adulthood [6]. The absence of normal menstruation is an indicator of infertility and should assess for pathology [7]. Positive correlations between the degree of adiposity and earlier pubertal development in girls have been reported. Conversely, females in whom leanness and strenuous physical activity have coexisted from early childhood frequently exhibit a marked delay in menarche and they frequently have oligomenorrhea or amenorrhea. These observations support the thesis that the energy balance is related to the activity of initiating puberty via hormonal signals. Onset of puberty is influenced by genetic, social, environmental, ethnicity and nutritional factors [1]. Similar findings showed an inverse association of age at menarche (AAM) with mean Body Mass Index (BMI) [8-12]. Obesity and a reduction in the age at menarche, result in the adverse effect on linear growth in girls. Everyone’s stature, will effect on mental health, social and economic status. Due to changes in AAM in the world, the aim of this study on 18 to 21 yr old medical students was to determine approximate AAM and its relationship to their weight, height and BMI at Mashhad University of Medical Sciences.
Materials and Methods
Data collection
This cross sectional study included 466 medical students aged 18-21 yrs old by convenient sampling in Mashhad Medical Sciences Azad University of Iran. We obtained data between September 2016 and November 2017 through questioner and interviews with medical students.
Questioner had acceptable validity and reliability. Anthropometric information included weight, height and BMI. Measurements were performed by two medical students. Height was measured with a portable height gauge instrument (SEGA) in a standing position with bare feet. Height measurements were calculated twice, and the mean value was considered. A portable scale sensitive to 0.1 kg was used for weight measurement at a similar medical scale (SEGA), which was conducted with minimal under clothe. A lot of care was taken to ensure the accuracy of the research and reliability tools.
The diagnostic criteria of the World Health Organization (WHO) were used to define BMI, overweight and obesity. BMI at the time of the screening examination was calculated as weight (kg) divided by the square of height (m2) in meters. To measure obesity, the body mass index (BMI) was used. BMI equal to 30 kg/m2 was considered as a threshold for obesity in women and men (≥ 95 Percentile of BMI) and overweight (Pre-obesity) was defined as BMI 25.0 to 29.9 kg/m2 with a percentile of 95>BMI ≥ 85, (9-12). AAM considered the onset of menstrual activity (menarche).
Study design
Medical students were screened for enrollments if they were healthy females, aged 18-21 yrs old. Pregnant women, people with diseases affecting their weight and height like diabet, hypothyroidism, chronic infectious diseases, anemia, drug abuser and psychological disorders were excluded.
From 480 students, 14 persons did not meet the inclusion criteria and were excluded. Out of 466 students, 5 were absent or refused the study and 461 students enrolled the study (Figure 1).
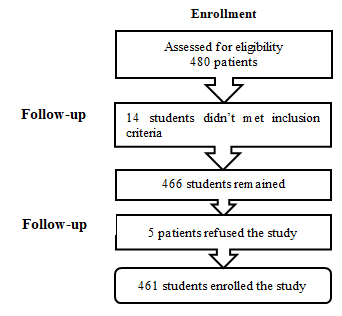
Figure 1: Consort Flow Diagram.
Medical students received questioner that included age, race, height, weight, BMI and age at menarche, parent`s job, parent`s level of education, socioeconomic status and age of the patients.
To describe data, statistical tables and indicators such as average has been used.
Statistical analysis
Data was expressed as mean ± standard deviation or percentages based on the characteristics of the variables.
Data analysis was expressed using the parametric method of Pearson’s correlation coefficient through SPSS, v.18. Statistical significance was assumed at p<0.05 with a confidence interval of 95%.
Results
There was no significant difference regarding to AAM and age, race, height, weight, BMI, parent`s job, parent`s level of education and socioeconomic status. P>0.05.
The minimum and maximum age at menarche was 6 and 18 yrs respectively. In 82.3% of students AAM was between 12 and 16 yrs.
The prevalence of early menarche, defined as less than 11 years of age, was 13.8% and in 3.9%, AAM was more than 17 yrs old (Table 1).
| (Age at menarche (yr) | ||||||||
|---|---|---|---|---|---|---|---|---|
| <11 | 12-16 | >17 | Frequency distribution | |||||
| Height Growth(meter) | N | % | N | % | N | % | N | % |
| <1.55 | 10 | 23.3 | 32 | 74.4 | 1 | 2.3 | 43 | 100 |
| 1.56-1.66 | 45 | 16.08 | 219 | 81.74 | 4 | 1.5 | 268 | 100 |
| >1.66 Total | 8 | 5.4 | 126 | 85.7 | 13 | 8.8 | 147 | 100 |
| 63 | 13.8 | 377 | 82.3 | 18 | 3.9 | 458 | 100 | |
| Pearsons R = 0 . 259 | P-value=0.0001 | |||||||
| Weight growth(kg) | N | % | N | % | N | % | N | % |
| <45 | 1 | 7.1 | 13 | 92.9 | 0 | 0 | 14 | 100 |
| 46-55 | 18 | 13.1 | 11.6 | 84.7 | 3 | 2.2 | 137 | 100 |
| 56-65 | 34 | 15.3 | 179 | 80.6 | 9 | 4.1 | 222 | 100 |
| 66-75 | 8 | 11.6 | 55 | 79.7 | 6 | 8.7 | 69 | 100 |
| >76 | 2 | 12.5 | 14 | 87.5 | 0 | 0 | 16 | 100 |
| Total | 63 | 13.8 | 377 | 82.3 | 18 | 3.9 | 455 | 100 |
| Pearson R=0 . 030 | P-value=0.519 | |||||||
| BMI,kg/m2 | N | % | N | % | N | % | N | % |
| <30 | 62 | 13.7 | 372 | 82.3 | 18 | 4 | 452 | 98.7 |
| ≥30 | 1 | 16.7 | 5 | 83.3 | 0 | 0 | 6 | 1.3 |
| 63 | 13.8 | 377 | 82.3 | 18 | 3.9 | 458 | 100 | |
| Pearsons R= 0.019 | p-value=0.681 | |||||||
Table 1: Average age of menarche based on weight and height growth and BMI in the Subjects.
BMI in 98.7% of the subjects was less than 30 kg/m2 (Table- BMI).
The mean age at menarche was 13.32 ± 1.78 years in students with BMI less than 30 kg/m2.
Early menarche at the age of less than 11 years in obese students was 16.7% and, in those with BMI less than 30 kg/ m2 was 13.7% but not significantly (Table-BMI, P>0.05).
The prevalence of obesity, defined as BMI ≥ 30 kg/m2, was 1.3%. There was no significant relationship between AAM and weight and BMI of the subjects, P>0.05.
There was no significant reversal relation between AAM and weight and BMI in this research. (Table-P>0/05). There was a significant positively correlation between AAM and height growth in studied medical students, (Table-height, P=0/0001). Height abundance of 1.56 to 1.65 meters was higher in the subject.
Discussion
Present research showed the mean age of menarche at 13.32 ± 1.782 yrs. The mean age at menarche is different in various cities in Iran and is reported at 12.6, 13.42 and 12.6, in Bandar Abas, Shahre Rey and Zanjan cities respectively [13-15].
In Tehran, Gorgan, Shahrud and Mazanderan, the mean age of menarche is reported at 12.55, 13.54, 12.9, and 12.5 yrs respectively [5,16-18]. In similar studies in the world the mean menarche age is revealed at 12.41, 12.54 yrs in Turkey and USA, respectively and 13.9 and 13.66 yrs, in Stockholm and Ghana respectively [19,20]. In a cross sectional survey enrolled in Indonesia, Most girls with a mean age of 15 yrs had reached menarche.
Menarche age was significantly higher in girls of fathers with lower education but there was no association between AAM and social status and parental occupation in Stockholm. These results were consistent with our report [18-20].
The mean menarcheal age of 13.32 yrs found in our study was above the average age at menarche reported 12.81 yrs in a survey of the meta-analysis by Bahrami et al. in Iran. In this meta-analysis, the mean age at menarche (12.81) was less than some European countries such as Switzerland, Sweden, and Denmark, more than some countries such as Greece and Italy, and similar to the findings in the United States of America and Colombia. Researchers conducted in different countries show that the lowest and the highest mean ages at menarche are for Thailand and England (11.2 and 13.5 yrs), respectively. In Iran the lowest and highest mean ages at menarche were reported in Sari (11.4) and Birjand (13. 8 yrs) cities respectively.
In Gorgan Ù? intermediate school students had menarche at an earlier age (12.20 ± 1. 45 yrs) compared to university students (13.54 ± 1.30 yrs), (P<0.05). The AAM of intermediate school students decreased by 68.54 days per year. Since a change in the living conditions of the industrial age at menarche decreases, this trend seems logical [17]. Compared to the first Ghanaian study in 1986, the decline per decade of menarcheal age of 0.11 yr was recorded. Declining mean menarcheal age is strongly correlated with improved socio economic development.
Similar study in Bangladesh revealed that more than 48% of girls had historical menarche age of 12 yrs, and the other girls experienced menarche at the age of 10 and 11 yrs. It was observed that significantly (p<0.01) more number of girls who lived in rich family reached menarche than that of comparatively poor family. Therefore, modern lifestyle changes may have the important factors for early age at menarche in Bangladesh. In Tokyo the mean menarcheal age had changed from 15.1 yrs in those born up to 1900 to 12.5 yrs in those born during the 1960’s.
Various researches show that the age at menarche had been decreasing in the 19th and 20th centuries and also in recent yrs. During the 20th century, puberty had been decreasing about 3 months per decade and also a year earlier for girls than in the 1970’s and the age of breast development which is the first clinical sign of female puberty has declined three months per decade between 1977 and 2013.
A review revealed that this variation in AAM is likely to increase the disease for women in: mental health, fertility related conditions, cardiovascular disease, and bone health, diabetes, hypertension, dyslipidemia, liver disease, lung disease, breast cancer, Bipolar Disorder and etc. A higher age specific BMI is contributing to early pubertal onset. But excess weight alone cannot explain this drastic shift. This results were consistent with our report. Age at onset of menarche did not accelerate even in a childhood population with more than 10% obesity prevalence.
In our study, Early menarche at the age of less than 11 yrs in obese students was 16.7% and, in those with BMI less than 30 kg/m2 was 13.7% but not significantly P>0.05. There was no significant relationship between AAM and weight and BMI (Table-P>0/05). Similar studies on Tehran, zanjane, Kuwait, Kish Isand of Iran, and some other studies reported that the girls with early menarche are more overweight [5,12,13].
In present study, height of 9.4% of the participants was less than 1.55 meters that was significantly according to history of their low menarche age. (The Table-Positive Pearson correlations). There was a significant positive relation between AAM and height growth in studied medical students, (Table- height, P=0/0001).
In a similar research in Iran, The height and weight averages of the studied children were more than the averages of standard growth charts. However, as age increased, the height and weight averages became less than the standard growth.
In a cross sectional study in Nigeria, there is a significant reverse association between menarche age and social class p=0.001. There have been no significant association between overweight and obesity with menargeal age P>0.5, this results were consistent with our report. There were significant relationships between AAM and height, BMI and the maternal educational level. The relation between AAM and weight and lipid profiles of subjects was not statistically significant. These results were consistent with our report.
In a similar study, the AAM wasn`t significantly related to somatic characteristics of the girls, but higher socioeconomic state was associated with lower age at menarche. A research conducted as review article during 1999 in Iran, reported AAM at 13.4 yrs, which comparing to our study, the AAM was higher than our research (13.4VS.13.32, respectively). The decline of AAM per 20 years in present study was 0.1 yr (1.2 months per 2 decade). In a similar study which reported menarche at a mean age of 11.71 yrs, adolescents with excess weight had an earlier menarche than their peers. These differences are due to various geographic locations, climate, environmental, racial BMI, biological, socioeconomial, exercise and cultural conditions. Also, AAM is influenced bby lifestyle, and genetic factors.
Limitations
This study was undertaken among medical female students due to limited resources. The results can therefore not to be generalized for all females in Iran. Measuring ages at menarche using retrospective or recall method is influenced by poor memory. The recall technique was the only feasible method.
Conclusion
The mean age of menarch in our study has been lower compared to previous study in about two decades ago, conducted at 1999, in Iran, (13.32 VS. 13.4 years), respectively.
Due to socioeconomic conditions and lifestyle changes in Iran, can be expected to decline the age of menarche over the coming decades.
Early menarche is one of the causes of short stature and various disease mentioned before. However it is necessary to plan intervention programs within families and school managers to improve dietary patterns and physical activity of this age group to postpone AAM and consider menstrual problems that can occur in an adolescent. In addition there is also a risk of sexual abuse and undesirable pregnancy. Further studies are needed to confirm these findings obtained in this study.
Ethical Approval
Prior approval for this study was obtained from the Ethics Committee of the School of Medicine and Health Sciences of the University for Development Studies. Informed consents for participation were obtained from all subjects about the study together with the invitation to participate. By returning the questioner, the woman agreed to participate.
ORCID IDs
Saeedeh Tarvij Eslami: https://orcid.org/0000-0003-0095-3031.
Hosain Nasirian: https://orcid.org/0000-0002-7756-0829.
Conflict Of Interest Statement
The authors declare that they have no competing interests.
Acknowledgements
We would like to express our gratitude to the office of Education of Medical science, Islamic Azad University for their assistance and cooperation during this research and to Medical students for their cooperation.
Authors Contributions
Hossein Nassirian: Implementation the study, manuscript preparation, principal investigation. Saeedeh TarvijEslami: Contributions to conceive the study, principal investigation, critical revision of the manuscript for important intellectual content, study conception, and manuscript preparation. Hamed Shafagh: Participation in the study implementation and manuscript preparation Maryam Gholami: Acquisition of data, manuscript preparation, statistical analysis. Kimia Vatani: Participation in the study implementation and manuscript preparation, submitting the article. All the authors read and approved the final manuscript.
Funding
None
References
- Frank B. Growth hormone plays key role in early puberty, breast cancer risk. J Adolesc Health. 2020.
- Anikwe CC, Mamah JE, Okorochukwu BC, et al. Age at menarche, menstrual characteristics, and its associated morbidities among secondary school students in Abakaliki, southeast Nigeria. Science Direct, Heliyon. 2020;6 (5), e04018.
- Cesario SK, Hughes LA. Precocious puberty: A comprehensive review of literature. J Obstet Gynecol Neonatal Nurs. 2007;36(3):263-74.
- Rabbani A, Motlagh ME, Mohammad K, et al. Assessment of pubertal development in Iranian girls. Iran J Pediatr. 2010;20(2):160-6.
- Yang X, Chen R, Zhang Y. Etiology and prognosis of peripheral precocious puberty in children. Zhongguo dang dai er ke za zhi Chinese J Contemp Pediat. 2011;13(12):947-50.
- Lacroix AE, Gondal H, Langaker MD. A Physiology, Menarche.Last Update: August 24, 2020. StatPearls.
- Farahmand M, Ramezani Tehrani F, Khalili D. Is there any association between age at menarche and anthropometric indices? A 15-year follow-up population-population-based cohort study. Eur J Pediatr,. 2020;179(9):1379-88.
- Rosenfield RL, Lipton RB, Drum ML. Thelarche pubarche, and menarche attainment in children with normal and elevated body mass index. Pediatrics. 2009;123(1): 84-8.
- Klishadi R, Hashempour M, Sarafzadegan N. Prevalence of overweight and obesity among guidance and high school student, and its relation to modifiable environmental factors. Tehran Univ Med J (TUMJ). 2003; 61:260-73.
- Irena Bralić, Tahirović H, Matanić D, et al. Associaton of early menarche age and overweight/obesity.J Pediatr Endocrinol Metab. 2012; 25 (1-2):57-62.
- Khoshnevisasi P, Sadeghzadeh M, Mazloomzadeh S. Age at Menarche and its Related Factors among School Girls, in Zanjan, Iran Int J Pediat, 2017;5(4), Serial No.40.
- Fazli A, Mousavi SM, Namazi SH. Puberty age in girls, Bandar Abbas, Iran (2000). Med J Hormozgan Univ. 2007; 11:65-9.
- Moghimi A, Razaghi Azar M, Ebrahim B. Puberty in the school girls of Shahre-e-Ray. J Arm Univ Med Sci I.R. Iran. 2003; 1:175-80 .
- Gharravi AM, Gharravi S, Marjani A. Correlation of age at menarche and height in Iranian student girls living in Gorgan-Northeast of Iran. JPMA J Pak Med Assoc. 2008;58(8):426-9
- Delvarianzadeh M, khosravi A, Bolbolhaghighi N. Relationship between anthropometric parameters with menarche age of school girls (11-14 years old) in Shahrud. Knowledge Health. 2008;3(3-4),43-7
- Delavar MA, Hajian-Tilaki KO. Age at menarche in girls born from1985-1989 in Mazandaran, Islamic Republic of Iran Eastern. Mediterranean Health J. WHO. 2020.
- Semiz S, Kurt F, Kurt DT. Factors affecting onset of puberty in Denizli province in Turkey. Turk J Pediatr. 2009;51(1):49-55.
- Anderson SE, Dallal GE, Must A. Relative weight and race influence average age at menarche: results from two nationally representative surveys of US girls studied 25 years apart. Pediatrics. 2003;111(4):844-50.
- Furu M. Menarcheal age in Stockholm girls.Ann Hum Biol.1976;3:587-90.
- Ameade EPk, Garti HA. Age at Menarche and Factors that Influence It: A study among female university students in tamale, Northern Ghana. PLoS One. 2016;11 (5): e0155310.
Author Info
Hossain Nassirian1, Saeedeh TarvijEslami1*, Hamed Shafagh1, Maryam Gholami2 and Kimia Vatani12Department of Internal Medicine, Bojnord University of Medical Sciences, Iran
Published: 31-Aug-2021
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.